 
Two core principles lay at the foundation of Dalton’s atomic theory: the indivisible nature of atoms and their unique chemical properties. Core Principles of Dalton's Atomic Theory This law, derived from Dalton’s study of weights and combinations of elements and compounds, provided compelling evidence for the existence of atoms and laid the groundwork for our understanding of the composition of chemical compounds. 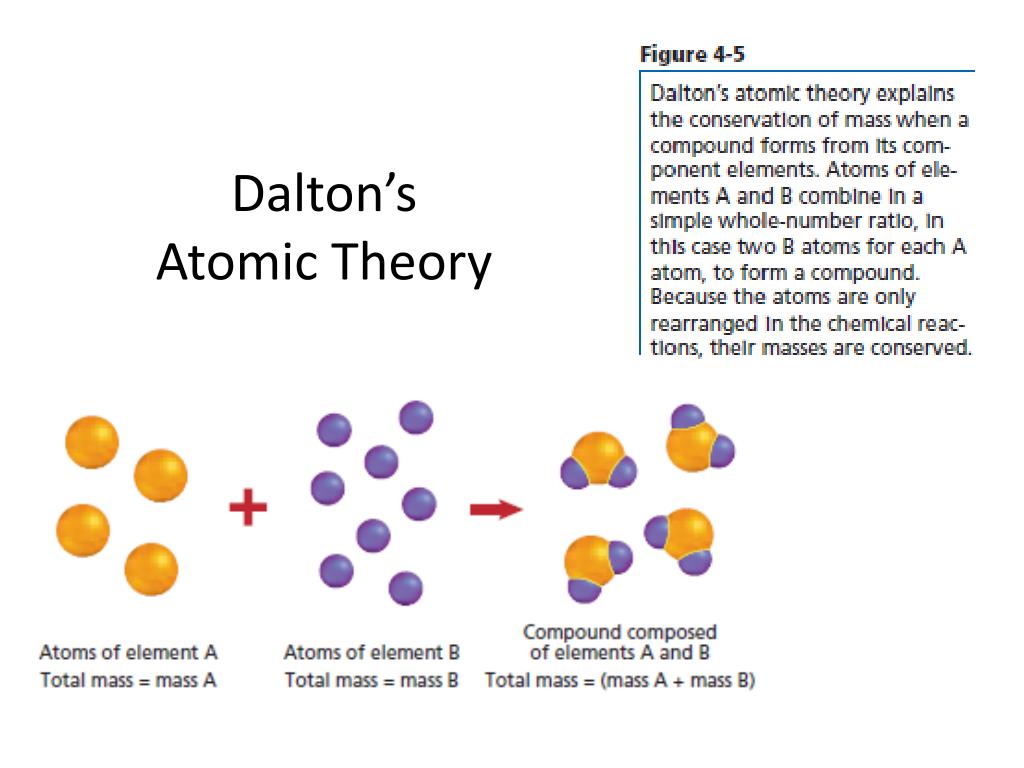
Dalton noted that when elements form more than one compound, the masses of one element combining with a fixed mass of the other are in the ratio of small whole numbers. The law of multiple proportions, a vital element of Dalton’s atomic theory, stands as evidence of Dalton’s detailed observations and logical deductions. Instead, he relied on his distinct experimental data and interpretations, thus laying a firm, independent foundation for modern atomic theory. He suggested that these atoms could neither be created nor destroyed, only rearranged, and that each element’s atoms were unique and varied in weight.ĭespite the abundant scientific inquiry during his time, Dalton’s theory was not directly shaped by his contemporaries. His theory posited that all matter consists of indivisible particles - atoms. Formulating the Theoryĭalton presented his atomic theory in 1804, backed by extensive research and experiments. These observations provided a foundation for his atomic theory and were instrumental in the formulation of the law of multiple proportions. Furthermore, he noticed a consistent pattern in the fixed ratios in which matter combined. His critical observations highlighted that the total pressure of a mixed gas equaled the sum of the partial pressures of each individual gas. Dalton's Observationsĭalton’s scientific journey was marked by his meticulous examination of how elements combined and interacted. Dalton hypothesized that atoms were the building blocks of all matter and proposed a scientific theory that would bridge the gap between philosophy and science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
